Abstract
Purpose
To identify factors which predict, among estrogen receptor (ER)-positive breast cancer patients, who chooses to take adjuvant tamoxifen.
Methods
We studied 1347 women with ER-positive breast cancer who were treated at Women’s College Hospital between 1987 and 2000. For each patient, we obtained information on age at diagnosis, tumour size, lymph node status, ER status, treatments received and dates of local recurrence and of death. We compared women who did and who did not take tamoxifen for a range of factors. We used the Kaplan–Meier method to estimate 15-year local recurrence-free and breast cancer-specific survival rates for women who did and who did not take tamoxifen.
Results
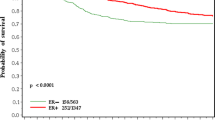
Overall, 50.4% of women who had a mastectomy took tamoxifen and 61.0% of women who had a lumpectomy took tamoxifen (p = 0.002). Tamoxifen use did not correlate with any of the factors that were predictive of a high risk of death from breast cancer, such as young age, large tumour size and positive lymph node status. Young women (<40 years) experienced much higher mortality (41.1%) than older (>60 years) women (14.1%, p < 0.01), but were much less likely to have taken tamoxifen (35.0 vs. 74.6%, p < 0.01).
Conclusions
Approximately one-half of women with ER-positive breast cancer who are candidates for tamoxifen did not take tamoxifen. Women with lumpectomy were more likely to take tamoxifen than women with mastectomy. Paradoxically, women at high risk of death from breast cancer (less than 40 at diagnosis and/or lymph node positive) and who are expected to receive the greatest benefit from tamoxifen in terms of mortality reduction were less likely to take it than were low-risk women (older women, lymph node negative). These findings suggest that women consider the reduction in risk from local recurrence to be more important than the reduction in risk of death from breast cancer when they consider taking tamoxifen.



Similar content being viewed by others
References
Mariotto A, Feuer EJ, Harlan LC, Wun LM, Johnson KA, Abrams J (2002) Trends in use of adjuvant multi-agent chemotherapy and tamoxifen for breast cancer in the United States: 1975–1999. J Natl Cancer Inst 94:1626–1634
Gradishar WJ, Anderson BO, Balassanian R et al (2016) Invasive Breast Cancer Version 1.2016, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw 14:324–354
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365:1687–1717
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), Davies C, Godwin J et al (2011) Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378:771–784
Hershman DL, Kushi LH, Shao T et al (2010) Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients. J Clin Oncol 28:4120–4128
Partridge AH, Wang PS, Winer EP, Avorn J (2003) Nonadherence to adjuvant tamoxifen therapy in women with primary breast cancer. J Clin Oncol 21:602–606
Friese CR, Pini TM, Li Y et al (2013) Adjuvant endocrine therapy initiation and persistence in a diverse sample of patients with breast cancer. Breast Cancer Res Treat 138:931–939
Grunfeld EA, Hunter MS, Sikka P, Mittal S (2005) Adherence beliefs among breast cancer patients taking tamoxifen. Patient Educ Couns 59:97–102
Llarena NC, Estevez SL, Tucker SL, Jeruss JS (2015) Impact of fertility concerns on tamoxifen initiation and persistence. J Natl Cancer Inst. doi:10.1093/jnci/djv202
Biglia N, Cozzarella M, Cacciari F et al (2003) Menopause after breast cancer: a survey on breast cancer survivors. Maturitas 45:29–38
Dorjgochoo T, Gu K, Kallianpur A et al (2009) Menopausal symptoms among breast cancer patients 6 months after diagnosis: a report from the Shanghai Breast Cancer Survival Study. Menopause 16:1205–1212
Jatoi I, Proschan MA (2005) Randomized trials of breast-conserving therapy versus mastectomy for primary breast cancer: a pooled analysis of updated results. Am J Clin Oncol 28:289–294
Veronesi U, Cascinelli N, Mariani L et al (2002) Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med 347:1227–1232
Stewart HJ, Prescott RJ, Forrest APM (2001) Scottish Adjuvant Tamoxifen Trial: a randomized study updated to 15 years. J Natl Cancer Inst 93:456–462
International Breast Cancer Study Group, Colleoni M, Gelber S et al (2006) Tamoxifen after adjuvant chemotherapy for premenopausal women with lymph node-positive breast cancer: International Breast Cancer Study Group Trial 13-93. J Clin Oncol 24:1332–1341
Cahir C, Dombrowski SU, Kelly CM, Kennedy MJ, Bennett K, Sharp L (2015) Women’s experiences of hormonal therapy for breast cancer: exploring influences on medication-taking behaviour. Support Care Cancer 23:3115–3130
Metcalfe KA, Esplen MJ, Goel V, Narod SA (2004) Psychosocial functioning in women who have undergone bilateral prophylactic mastectomy. Psychooncology 13:14–25
Clarke M, Collins R, Darby S et al (2005) Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet 366:2087–2106
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional and/or National Research Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Narod, S.A., Sopik, V. & Sun, P. Which women decide to take tamoxifen?. Breast Cancer Res Treat 164, 149–155 (2017). https://doi.org/10.1007/s10549-017-4226-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4226-4